How to get potential mass spec fragments on chem draw
The identity of the isotope marker for the terminal base is then determined by combustion, and mass spectrometric analysis of the resulting isotopically labeled sulfur dioxide. The nucleotide fragments are separated by capillary electrophoresis.
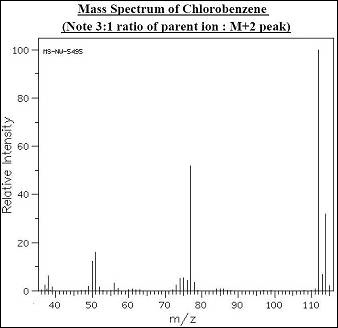
Specifically, the four stable isotopes of sulfur (32, 33, 34, 36) will be used to identify the four terminal bases (A, T, G, C ) where the label is incorporated as a thiophosphate bridge. The method is based on a mass spectrometric determination of the four component terminal nucleotide residues, where the information regarding the identity of the individual nucleotides is contained in the mass of a stable nuclide marker. Preliminary work on a new approach to sequencing polynucleotides by the Sanger procedure is presented. If you cant get the whole structure put as many fragments as you can. These peptides are then broken into fragments and these fragments are analyzed in a second mass analyzer they in the second mass spec stage. And then, based on this you select certain ions with a very particular m over z value for further analysis. New methods to sequence DNA by mass spectrometry New methods to sequence DNA by mass spectrometryīrennan, Thomas Chakel, John Bente, P. The relative abundance of an isotope is the percentage of atoms with a specific atomic mass found in a naturally. The 1H and 13C NMR spectra of a compound with chemical formula C 4 H 6 O 2 are. And, in the first mess, mass spec the ions are separating separated according to their mass to charge ratio, m to z.
